In the Journal of the American Chemical Society, Stylianou et al have reported on the investigation of the structural transformation of a Zn-based flexible
Recent MOF Publications
Metal-organic frameworks (MOFs) are crystalline inorganic-organic solids formed from metal ions or clusters linked by organic bridges. MOFs have promise as materials for a variety of applications, including gas storage, purification and separation, as well catalysis and molecular sensing.
Here we highlight two recent publications where Hiden Isochema instruments have been used to characterize metal-organic frameworks (MOFs).
MOF synthesis
Capturing chemical intuition in synthesis of metal-organic frameworks
M. Moosavi, A. Chidambaram, L. Talirz, M. Haranczyk, K. C. Stylianou and B. Smit
Nature Communications (2019) 10, 539. DOI: 10.1038/s41467-019-08483-9
The synthesis of metal-organic frameworks (MOFs) is dependent on often subtle variations in conditions, which are generally determined experimentally but is also by the chemical intuition of synthetic chemists. The authors of this study suggest that machine learning from a body of both successful and ‘failed’ or partially successful syntheses can be used to predict the optimal synthesis conditions more efficiently. As a case study, robotic synthesis of MOF HKUST-1 was performed using a range of different conditions and the resulting material characterised using analytical methods including gas sorption using an IGA-001 to determine the BET surface area, which is used as an initial indicator for a successful synthesis. The results are used to provide “learnt chemical intuition” which was then applied to refine the synthesis of HKUST-1 with the highest yet reported surface area. The learnt chemical intuition was then applied to synthesis of a related but different MOF to demonstrate how synthesis could be significantly streamlined in the future.
MOF flexibility
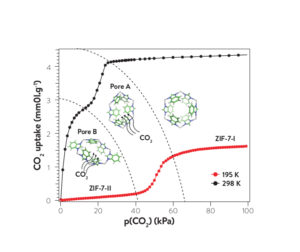
Structural dynamics of a metal–organic framework induced by CO2 migration in its non-uniform porous structure
P.Zhao, H. Fang, S. Mukhopadhyay, A. Li, S. Rudić, I. J. McPherson, C. C. Tang, D. Fairez-Jimenez, S. C. E. Tsang and S. A. T. Redfern
Nature Communications (2019) 10, 999. DOI: 10.1038/s41467-019-08939-y
A recent study describes CO2 adsorption by a flexible MOF, ZIF-7, and the effect on the structure of the MOF. The authors used an IGA-001 to measure high resolution CO2 sorption isotherms at several temperatures, and a number of X-ray and neutron based methods to evaluate the structural dynamics of the material. The authors conclude that the MOF’s structural change, induced by CO2 adsorption and exhibited by an abrupt increase in CO2 sorption capacity between 40 and 60 kPa at 298 K, is due to CO2 migration rather than a pore opening mechanism. CO2 sorption at 195 K exhibits a two stage adsorption process, the second of which is correlated with the ZIF-7 phase change. The authors comment that the dynamics observed for the CO2 / ZIF-7 system provides a new insight into the phenomenon of MOF flexibility.
Hiden Isochema instruments have been used extensively to study the adsorption properties of zeolites, MOFs, and other nanoporous materials. The IGA-001 gravimetric analyzer can determine single component gas adsorption, while the IGA-002 can be used for vapor sorption and theIGA-003 and IGA-100 offer gas and vapor mixture capability. XEMIS models such as the XEMIS-001 and XEMIS-002 provide similar function to the equivalent IGA models, with higher pressure operation and compatibility for corrosive species. The IMI-FLOW can be used for flowing gas mixture measurements with a close-coupled quadrupole mass spectrometer from Hiden Analytical, while the ABR is dedicated to the measurement of breakthrough curves for gas separation studies.
Please contact us for more information on any of product range or for a quotation.


